This page is a copy of a complaint filed with the Georgia Composite Medical Board.
MEDICAL ETHICS COMPLAINT REGARDING A MEDICATION INDUCED DVT TO PATIENT BARBARA MORGAN HICKSON 2012/2013
After very careful consideration, I submit this complaint to the Georgia Composite Medical Board as an ethics complaint against the following 4 physicians who practiced at the Mayo Clinic Health System in Waycross, GA during 2012 and prescribed (in the order indicated below) the medication Megace to my geriatric mother; the late Barbara Morgan Hickson:
Dr Ashraf I. Sedfawy, MD
Dr Paresh Thanki, MD
Dr John F. Michaels Jr MD
Dr Jules M. Nehmetallah, MD
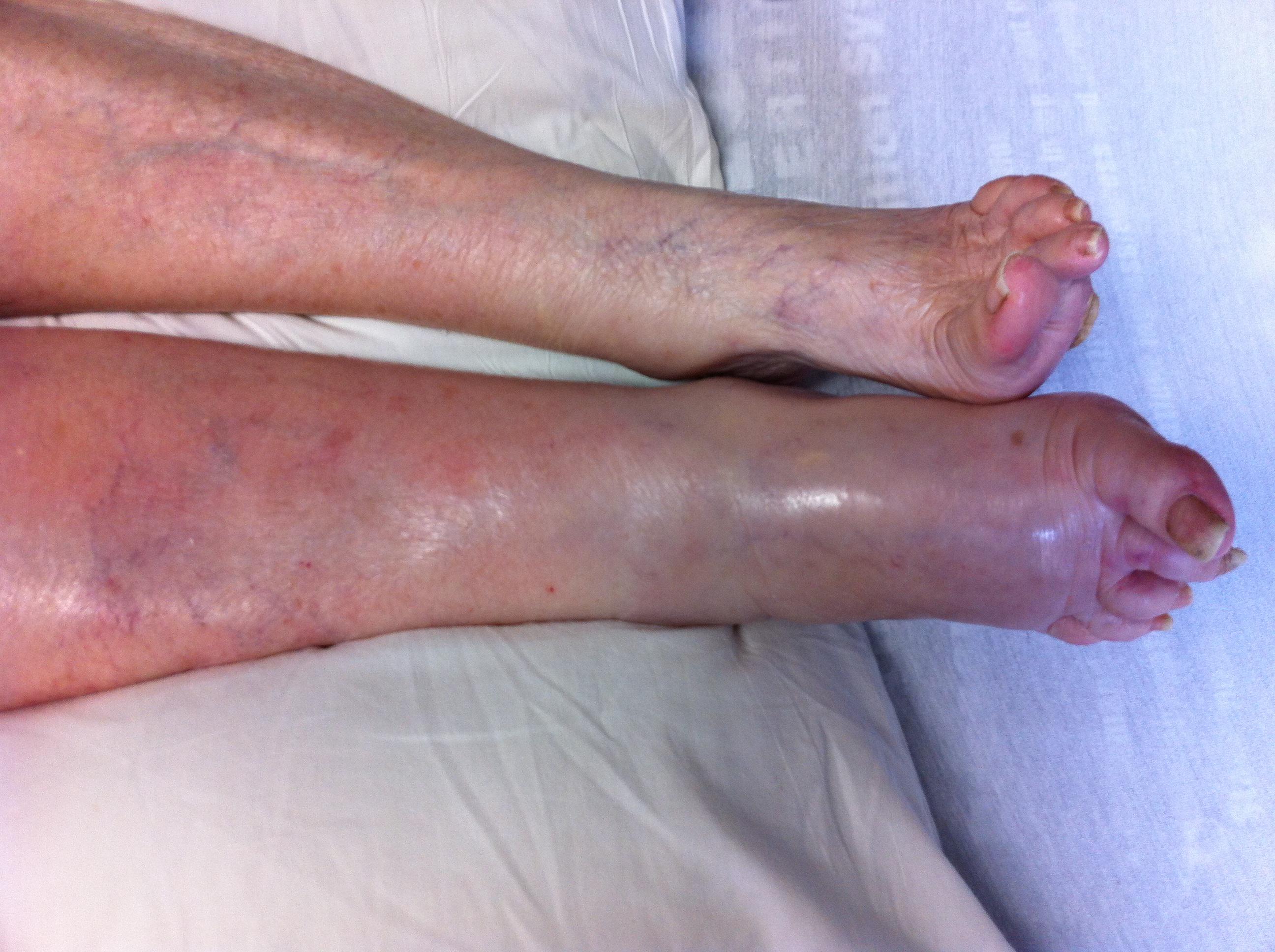
BACKGROUND
During the period April 2012 through June 2013 my geriatric mother age 79-80, Barbara Morgan Hickson, was in and out of the Mayo Clinic Health System hospital in Waycross. The series of hospital visits began with her suffering a fall at home, which resulted in a compound fracture to her right arm. She was given a general anesthesia and subsequently started displaying signs of postoperative cognitive dysfunction which was diagnosed as “dementia” in hospital records. Additionally, it was noted early on that she displayed a significant loss of appetite.
Following the repair of her broken arm, she was discharged from the hospital into the care of Baptist Village Nursing home in Waycross for rehabilitation. During this time the physician responsible for patients residing at Baptist Village was Dr Donald Richard Lynch, MD of Waycross. After her admittance to Baptist Village her husband, Mark L. Hickson, decided to switch primary care doctor from Dr. Ricardo Zarzuela of Waycross to Dr Donald R. Lynch also of Waycross. One of the early recommendations made by Dr Lynch to Mr Hickson was that he have a Waycross psychiatrist examine Mrs Hickson to determine if any psychiatric conditions, such as depression, could account for her lack of appetite. Also, a number of Mayo Clinic Waycross hospital record notations state that her husband repeatedly made statements like “she's talking crazy” as the basis for entering medical statements about “psychosis” into the hospital records.
During this time I visited my mother for one week from my home in Virginia and learned that she was refusing to eat. On Friday May 11, 2012 I had a disagreement with her husband who wanted to have her admitted to the Mayo Clinic Waycross Senior Behavioral Unit (SBU) due to her lack of appetite. I had to leave Waycross on May 12th and it appears that her husband quickly took advantage of this fact and had her moved to the SBU. A family medical situation in Virginia required me to return home the weekend of May 12th to assist my wife with minor surgery that was accomplished on May 14th. During the week of May 14th, while back in Virginia, I began to research medications that might be available to stimulate appetite among geriatric patients. I discovered a number of promising candidates that were under examination within the medical research arena but surprisingly I did not find any drugs whatsoever that met the quality standard requirements applicable to my mother's case. Namely, that she was in a non-life threatening medical condition but there was great concern that all possible causes of her appetite loss should be examined quickly and a solution found. I opposed her admission to the SBU because I did not believe that other causes of appetite loss (e.g., swallowing and digestive track issues) had been evaluated. By skipping directly to psychiatric care I was concerned that psychotropic medications might make a proper diagnosis more difficult to obtain.
On May 18, 2012 I spoke via telephone from Virginia with Dr Paresh Thanki, MD a Waycross Psychiatrist in the SBU of the hospital. During this discussion Dr Thanki mentioned to me that he had placed my mother on the drug Megace for the purpose of stimulating her appetite. Since I had already read the available information regarding Megace I immediately raised an objection to my mother receiving this drug. I informed Dr Thanki that I had already researched the drug Megace and had ruled it out as a good candidate for my mother for the following 3 reasons:
-
Megace was NOT approved by the FDA for geriatric patients.
-
The only clinical trial I found that had been conducted for Megace involving geriatric patients was a single very small study. In this small study only 10% of geriatric patients given Megace reported any improvement. Thus, 90% were not helped by the drug.
-
The precautions/warnings indications for Megace from the manufacturer identified risks for Deep Vein Thrombosis (DVT) in patients with prior blood clot histories. I informed Dr Thanki that my mother had a history of DVT's dating back to the 1960's; from which she almost died.
To these objections, Dr Thanki replied: “Well what should I do then, do nothing?” I informed Dr Thanki that it was not my role to recommend any medical course of action but that I did want him to take my mother off of the drug Megace. I mentioned feeding tube or hydration or any other commonly used medical solutions just to get him to start thinking of sound alternatives. I also asked if any GI tract and/or swallowing tests had been conducted on her. At the conclusion of our conversation I was fully convinced that I had made myself clear on the Megace issue.
I arrived back in Waycross from Virginia on May 20th and immediately went to visit my mother in the SBU. I asked the charge nurse to confirm that she was off Megace. To my surprise the nurse informed me that she was still receiving Megace. I immediately asked who was on call that could change this and she provided me contact information for Dr John F. Michaels Jr MD. I phoned Dr Michaels and he contacted me back promptly. I discussed with him the conversation I had on May 18th with Dr Thanki and expressed great disappointment that my request to have Megace withdrawn had not been followed. Dr. Michaels assured me that it was a common practice in Waycross to administer Megace to geriatric patients who suffered from poor appetites and that the drug had helped many. I went over my 3 reasons for requesting that Megace be withdrawn especially emphasizing my mother's history of DVT's. Repeatedly, Dr Michaels emphasized that this would be a medical decision made solely on direction from a family member; essentially an action Against Medical Advice (AMA). Repeatedly, I continued to emphasize that I fully understood the significance of overriding physician ordered treatments but that I insisted that Megace be withdrawn. Given the level of resistance I was encountering to withdrawing Megace I became concerned that perhaps I did not understand the seriousness of her condition. I then asked Dr Michaels if my mother's medical condition was terminal? He replied no. Dr Michaels then spoke with the charge nurse and ordered the withdrawal of Megace.
Monday, May 21, 2012 was my mother's 79th birthday. I took balloons and a stuffed teddy bear to her room in the SBU. Later that morning I met with Dr Thanki, several nurses, and dietary representatives regarding my mother. Dr Thanki continued to express his belief that Megace was my mother's best course of treatment but we also discussed a number of her other medications and reached joint agreement to make a change in the medication he was using to treat her depression.
On May 22, 2012 my mother was transferred out of the SBU and back into the main hospital. Her indications were listed (See Annex A) as acute renal failure and psychosis. At this time, renal failure became the major focus and efforts were undertaken by Dr Nirmala D. Amaram, MD whose efforts continued for several weeks leading to a successful stabilization of her kidney function. The treatment and care provided to my mother by Dr Amaram was world class and I sincerely thanked him for his services. Not really understanding the importance of health care quality, her husband decided to replace Dr Amaram for subsequent treatments due to his sole stated reason that “he is not our kind of people;” a racial slur against Dr Amaram's Indian heritage.
On two separate occasions, two days after she reentered the regular hospital and again approximately 7 days after her admission, I asked for and received a print out of all the medications that my mother was taking. I did this solely for the purpose of insuring that she was not being given Megace. After the second report, I began to relax about Megace and to focus on the renal failure issues. I also again asked if any upper GI tests had been conducted to determine if there were any physical reasons for her lack of appetite. Necessity required me to return to Virginia for two weeks.
When I returned to Baptist Village Waycross I found my mother was moaning rhythmically and not drinking any fluids in addition to not eating. Her husband was present and I asked him about the moaning. “She does that quite often at home, lies in the bed moaning and after a few days is better” was his response. I told him that I thought the moaning indicated that she was experiencing some type of pain and that she should be see at the Mayo Clinic hospital. It required a few hours to convince him regarding my concerns but eventually she was taken by ambulance to the hospital where she was diagnosed with dehydration and a Urinary Tract Infection (UTI). She was rehydrated and discharged back to Baptist Village. After a few weeks I returned to my home in Virginia again for a few weeks and then in early May returned again to Waycross.
The Mayo Clinic Waycross hospital record dated 04/19/2012 (See Annex B) “Patients History” “Medications” section states: “Her primary care physician is Dr. Ric Zarzuela.” This statement makes it clear that the hospital knew what doctor to contact to obtain a copy of her medical records, which would have confirmed her history of DVTs.
Following the surgery for her compound fracture, Mrs Hickson awoke and demonstrated symptoms of postoperative cognitive dissonance. Thus, what she was able to communicate about her condition was quite limited. Her husband may/may not have known about her history of DVTs but he certainly had no knowledge or experience in evaluating a specific medication for use by a patient. Physician compliance with AMA ethical standards required that, at a minimum, a staff member from Mayo Clinic Waycross should have been instructed to contact Dr Zarzuela's office to request a fax copy of Mrs Hickson's medical records from her primary care physician. After Mrs Hickson's fractures were set she was released to Baptist Village for rehabilitation.
The Mayo Clinic Waycross hospital record dated 04/23/2012 (See Annex C) written by Dr Odette A. Thomas titled “Patient's History” shows that Mrs Hickson was brought back to the hospital on 04/23/2012 in an “altered mental status.” Her husband stated that she was hallucinating and not eating. The Plan section of this record states “6. Deep venous thrombosis and gastrointestinal prophylaxis.” Again, there is no indication in this record of any plan to have hospital staff contact her primary care physician Dr Zarzuela's office for information related to item 6. This should have been a customary reasonable preventive care procedure.
On my visit to Waycross in early May I again found that my mother was still not eating adequately or drinking fluids. I again insisted that she be returned to the hospital for examination. For this visit the diagnosis was again dehydration but acute kidney injury was also diagnosed. I did not accompany my mother into the examination room at the hospital so I was unaware of actions discussed or taken beyond hydration efforts. However, the Mayo Clinic Health record dated May 10, 2012 (See Annex D) contains a narrative summary by Dr Ashraf Sedfawy, MD which states:
“She is awake but she is just not hungry which is really nothing that we can change, it comes with age and the fact that she is a nursing home resident and we cannot reverse that. I will give her some Megace upon discharge to help with that, but I am not really expecting much.”
Since I was not present in the examination room I was unaware of any discussion to prescribe the drug Megace for my mother. Neither the doctor nor her husband informed me of this decision. There appears to be no indication in the Mayo Clinic Waycross records prior to this date of anyone contacting her primary care physician, Dr Ric Zarzuela regarding her medical history. The facts were that her history of multiple prior DVTs alone should have completely disqualified her from receiving this drug. This record reflects a serious medical misjudgment on the part of Dr Sedfawy who had insufficient medical history to prescribe this drug to Mrs Hickson. The administration of Megace to this geriatric patient violates:
-
The Georgia Composite Medical Board Rule 360-3-.02 Unprofessional Conduct Defined, in accordance with O.C.G.A.§§ 43-34-8 and 43-1-19, authorize the board to take disciplinary action licensees for unprofessional conduct as follows: (15) Failing to use medications and other modalities based on generally accepted or approved indications, with proper precautions to avoid adverse physical reactions, habituation, or addiction in the treatment of patients. However, nothing herein shall be interpreted to prohibit investigations conducted under protocols approved by a state medical institution permitted by DHS and with human subject review under the guidelines of the United States Department of Health and Human Services.
(15) Failing to use medications and other modalities based on generally accepted or approved indications, with proper precautions to avoid adverse physical reactions, habituation, or addiction in the treatment of patients. However, nothing herein shall be interpreted to prohibit investigations conducted under protocols approved by a state medical institution permitted by DHS and with human subject review under the guidelines of the United States Department of Health and Human Services.
(16) Failing to maintain patient records documenting the course of the patient's medical evaluation, treatment, and response.
2. The American Medical Association (AMA) Opinion 8.06 Prescribing and Dispensing Drugs and Devices which states:
“Physicians should prescribe drug, devices, and other treatments based solely upon medical considerations and patient need and reasonable expectations of the effectiveness of the drug, device or other treatment for the particular patient.”
At the time of this decision, Megace had not been approved for use in geriatric patients. There was inadequate scientific evidence (clinical trial data) for the FDA to approve the drug for geriatric patients and thus there could be no reasonable basis for any expected effectiveness of the drug for Mrs Hickson. The Dr Sedfawy was not involved in any Megace investigations under protocols approved by a state medical institution permitted by DHS under the guidelines of the United States Department of Health and Human Services. Further, Dr Sedfawy had not conducted any tests to rule out possible physical causes (e.g., upper GI, etc) of her condition and thus the drug therapy was premature. Lastly, Mrs Hickson's long history of DVTs should have been considered thus ruling out the prescription of Megace to her.
The Department of Justice has proven in court (April 2013) that Megace drug manufacturers were guilty of civil and criminal activities related to off-label-marketing of Megace to the medical profession for use in geriatric patients. The illegal activities of drug manufacturers, however, does not give physicians shelter to ignore either the AMA medical ethical standards nor Georgia laws and GCMB rules.
In his enthusiasm to rapidly assist Mrs Hickson with her appetite issues Dr Sedfawy tossed all reasonable caution to the wind by ignoring the absence of FDA approval for this medication for geriatric patients. He further failed to follow safe medical practices by not insuring that all treatable causes of weight loss (including GI tract obstructions) had been eliminated in accordance with the General Precautions issued by manufacturers of Megace. Lastly, he failed to properly vet Mrs Hickson for DVTs prior to prescribing Megace. As far back as September 12, 2000 Medscape (See http://www.medscape.com/viewarticle/413378, September 12, 2000) had published an article by Susan Russell Neary, PhD concluding that Megace should not be given to geriatric patients who had an underlying predisposition to thromboembolic events. The only thing that can be said in favor of Dr Sedfawy with regards to these violation of AMA ethics rules is he was not alone because each and every one of his mistakes were repeated by three other equally qualified physicians at Mayo Clinic Waycross.
On May 22, 2012 Mayo Clinic Waycross records (See Annex E) document an operative report by Dr Ahmad Irfan, MD. The purpose of the procedure was to rule out gastroparesis. A number of biopsies were taken to rule out various diseases. This procedure revealed a moderately large hiatal hernia. A distal esophageal structure was also noted. The esophagus was tortuous and appeared to have spasms. The patient's esophagus was dilated with a Maloney dilator, size 60 French. Following this procedure, Barbara Morgan Hickson began to display an modest increase in appetite. She was not eating full meals but did slowly show more interest in eating especially when, as is documented in hospital records, her son went to local restaurants and purchased take-out orders for her to eat instead of hospital food. Due to the fact that his approach worked nutritional notes were added to her hospital record stating that she could have any type of food she wanted prepared by the hospital.
On May 31, 2012 an Registered Nurse entered my moms hospital room at Mayo Clinic Waycross. While she went about her duties she and I told her about my increased optimism regarding her appetite. To my surprise, the nurse said: “Well her appetite should be improving from the Megace she is receiving.” Stunned, I informed the nurse that I had personally verified twice that my mother was not taking Megace. I followed the nurse back to the nursing station where she informed me that Dr. Jules M. Nehmetallah, MD has prescribed Megace for her. I immediately informed the nurse about my objections against my mother taking Megace and urged her to contact the doctor at once and advise him to terminate the prescription of Megace. Scattered throughout dozens of medical records (SEE Annex F For Example) for Barbara Morgan Hickson at Mayo Clinic Waycross subsequent to May 31, 2012 the following statement is written:
“Coded Allergies: MEGESTROL ACETATE (Verified Adverse Reaction, Intermediate, PT IS NOT ALLERGIC...SON REFUSES TO LET HER TAK. 5/31/12) DR NEHMATALLA JUST MARKED IT AS SUCH TO AVOID A SCENE...”
3. AMA Opinion 10.01 Fundamental Elements of the Patient-Physician Relationship states:
“The patient-physician relationship is of greatest benefit to patients when they bring medical problems to the attention of their physicians in a timely fashion, provide information about their medical condition to the best of their ability, and work with their physicians in a mutually respectful alliance.”
Based on the referenced note contained throughout Mayo Clinics records it is evident that Dr. Nehmetallah does not support this ethical standard. Further,
4. AMA Opinion 10.01 Fundamental Elements of the Patient-Physician Relationship states Item (2) states:
“The patient has the right to make decisions regarding the health care that is recommended by his or her physician. Accordingly, patients may accept or refuse any recommended medical treatment.”
Dr. Nehmetallah did not recommend the administration of Megace for my mother to either her husband or myself. Dr Nehmetallah simply felt it was the right solution for her and wrote the prescription without consulting any family member. This action is completely contrary to AMA ethical guidelines. I believe that failure to follow AMA's ethical guidelines, in this instance, cost my mother the pain and suffering of a DVT that she would carry for more than a full year and take with her to the grave.
The June 7, 2012 Mayo Clinic Waycross Record Consultation Report (See Annex G) written by Sridhar Srinivasan, MD to Dr. Aristotle P. Cochen, MD states:
“REASON FOR CONSULTATION: Deep venous throbosis
“She apparently has a history of deep venous thrombosis in 2010, and was treated by Dr. Zarzuela with Coumadin for about 6 months”
After months of me telling this to one Mayo Clinic Waycross doctor after the next, after explaining my concerns to one nurse after the next, after saying that an upper GI probe and other tests associated with physical causes for diminished appetite should be conducted at long last the necessary information from her primary care physician is finally documented in her hospital records!
On June 12, 2012 I asked for a face-to-face meeting with relevant representatives of Mayo Clinic Waycross to discuss what I believed to be the misuse of Megace by physicians and the DVT, which resulted from their actions of ignoring standard medical ethics practices. This meeting included Holly Sweat (Chief nursing officer), Randy Wheeler (Operations), Dr. Jules Nehmetallah, MD and staff. During the meeting Dr Nehmetallah stated that scientifically my mother had only a 2% chance of developing a DVT from Megace. I have no idea what study source Dr Nehmetallah was referencing but I do not believe that there is a shred of scientific evidence to date that an geriatric patient with a prior history (multiple occasions) of DVT's is only at a 2% risk from Megace. This is “voodoo science” and contrary to the findings of the Food & Drug Administration and to the civil and criminal guilty pleas Megace manufacturers entered with the Department of Justice on March 5, 2013.
CONCLUSIONS
The American Medical Association (AMA) establishes ethical standards for the medical community, which seek to balance the best interests of patients and the physicians. The conservative “do no harm” principal recognizes that the risks of allowing physicians to practice “rodeo medicine” are far too great to the general public. For these reasons, very stringent procedures and processes govern the conditions of clinical trials and other situations where there are elevated drug risks to patients. The Food and Drug Administration (FDA) expends a significant amount of its publicly funded (FY 2012 budget of $4.36 billion dollars) budget to insure that only medicines that have demonstrated (via scientifically controlled clinical trials) benefits for particular populations are approved for use by physicians. In spite of these safeguards, and without regard to being informed that Mrs Hickson had higher than average risk factors for DVT, four doctors at Mayo Clinic rushed forward one after the next until eventually she did develop a DVT. Shockingly, even after she developed a DVT hospital records in 2013 until her eventual terminal discharge continued to reflect Dr Jules Nehmetallah's opinion that he only withdrew Megace to “avoid making a scene.” Why then did the Department of Justice “make a scene” by finding the manufacturers of Megace guilty of civil and criminal law?
Off-label-drug prescriptions are not a rare occurrence in medical practice. There are numerous cases where the conditions of the patient do warrant throwing all caution to the wind and reaching for any experimental drug that could possibly have benefit. Especially when reaching for off-label purposes, it is incumbent upon the physician to have insured all due diligence with regard to considering the patients prior history in association with known drug side effects and to resort to high-risk practices only after all alternative causes of illness and treatments have been considered and ruled out. That was not done in this case.
This complaint documents serious allegations of ethical violations by four physicians at the Mayo Clinic Health Facility in Waycross, GA. The Georgia Composite Medical Board has a difficult job. It must balance the needs of the state, in terms of competent medical physicians, while protecting the health of Georgians through the objective enforcement of the Medical Practice Act of 2009. This includes, but is not limited to Items 7 and 22 of § 43-34-8(7), which state:
(7) Engaged in any unprofessional, unethical, deceptive, or deleterious conduct or practice harmful to the public, which conduct or practice need not have resulted in actual injury to any person. As used in this paragraph, the term "unprofessional conduct" shall include any departure from, or failure to conform to, the minimum standards of acceptable and prevailing medical practice and shall also include, but not be limited to, the prescribing or use of drugs, treatment, or diagnostic procedures which are detrimental to the patient as determined by the minimum standards of acceptable and prevailing medical practice or by rule of the board.
(21) Failed to comply with federal laws and standards relating to the practice of medicine or other health care profession regulated under this chapter, the regulations of drugs, the delivery of health care, or other related laws;
The four physicians identified in this complaint failed to comply with Georgia Composite Medical Board Rule 360-3.02 Unprofessional Conduct Defined. Amended (Referencing O.C.G.A. §§ 43-34-8 and 43-1-19), which states:
“(14) Failing to use such means as history, physical examination, laboratory, or radiographic studies, when applicable, to diagnose a medical problem.”
by neglecting to acquire the patients history from her primary care physician Dr. Ric Zarzuela and by ignoring her sons statements regarding the inappropriateness of Megace considering her prior history of DVTs and the Megace drug manufacturers precautions.
Further, the four physicians identified in this complaint failed to comply with Georgia Composite Medical Board Rule 360-3.02 Unprofessional Conduct Defined. Amended (Referencing O.C.G.A. §§ 43-34-8 and 43-1-19), which states:
“(15) Failing to use medications and other modalities based on generally accepted or approved indications, with proper precautions to avoid adverse physical reactions, habituation, or addiction in the treatment of patients.”
Where Megace was not approved by the FDA nor did it have clinical trials that scientifically demonstrated Megace to be safe or effective for geriatric patients; especially those with prior histories of DVTs.
Additionally, that on On June 12, 2012 in a meeting with other hospital staff present (see description provided above) that Dr. Jules Nehmetallah violated
Georgia Composite Medical Board Rule 360-3.02 Unprofessional Conduct Defined. Amended (Referencing O.C.G.A. §§ 43-34-8 and 43-1-19), which states:
“(19) Providing a false, deceptive or misleading statement(s) as a medical expert.”
when during the meeting described Dr Nehmetallah stated that my mother had a “2% risk of DVT” from Megace. There is no scientific basis whatsoever for Dr Nehmetallah's assertion that a patient with a history of multiple documented cases of DVTs had only a “2% risk” of acquiring a DVT as a result of being prescribed Megace. This erroneous “factual” assertion was made for the purpose of dismissing assertions by her son that the prescription of Megace to an elderly patient with a prior history of DVTs was, at the very least, unethical and unsound medical practice. Indeed, the Department of Justice had to intercede in the matter to prevent Megace drug manufacturers from promoting this medication with unproven results to physicians who were practicing irresponsible medicine.
That all four physicians violated Georgia State Board of Pharmacy Regulations 480-13-.09 Investigational Drugs. Amended which states:
“Investigational drugs shall be administered in accordance with an approved protocol that includes any requirements for a patient's appropriate informed consent.”
whereby all four physicians administered Megace without any FDA use approval, no clinical trials demonstrating safety and effectiveness of Megace for geriatric patients, and without properly vetting the patient to insure that her prior medical history qualified her to participate in an non-scientific experiment without patient or family consent.
Thus, to insure the safety of geriatric patients throughout the state a public reprimand, at a minimum, as provided for in § 43-34-9, of these four physicians should be documented and published by the Georgia Composite Medical Board in association with a full investigation of the facts and publication of the findings. Physicians who practice medicine that has no scientifically proven basis should do so with full public disclosure to their existing and future patients. This appears to be the best mechanism available to save lives and prevent unnecessary pain and suffering.
In conclusion, I sincerely hope that my efforts will result in additional protections and public admonishment of physicians who would knowingly place geriatric patients in the position of serving as “medical lab mice” without their informed consent and without the informed consent of their family members. A large portion of that responsibility ultimately rests on the shoulder of the Georgia Composite Medical Board.

Barbara Jean Morgan Hickson
